


Current Biotechnology ›› 2025, Vol. 15 ›› Issue (6): 952-959.DOI: 10.19586/j.2095-2341.2025.0146
• Reviews • Previous Articles Next Articles
Yujing WANG1( ), Jia WU1, Honge LI1, Shanshan LI1, Xiaoni YANG1, Qingqing GUO1, Wenran HU2(
), Jia WU1, Honge LI1, Shanshan LI1, Xiaoni YANG1, Qingqing GUO1, Wenran HU2( )
)
Received:2025-10-24
Accepted:2025-11-20
Online:2025-11-25
Published:2026-01-04
Contact:
Wenran HU
CLC Number:
Yujing WANG, Jia WU, Honge LI, Shanshan LI, Xiaoni YANG, Qingqing GUO, Wenran HU. The Ecological Niche Protection Function Mediated by Microbial Secondary Metabolites[J]. Current Biotechnology, 2025, 15(6): 952-959.
王玉晶, 吴嘉, 李红娥, 李珊珊, 杨晓旎, 郭清青, 胡文冉. 微生物次级代谢产物介导的生态位保护功能[J]. 生物技术进展, 2025, 15(6): 952-959.
| 群体感应信号 | 信号分子结构 | 微生物 |
|---|---|---|
| AHLs | 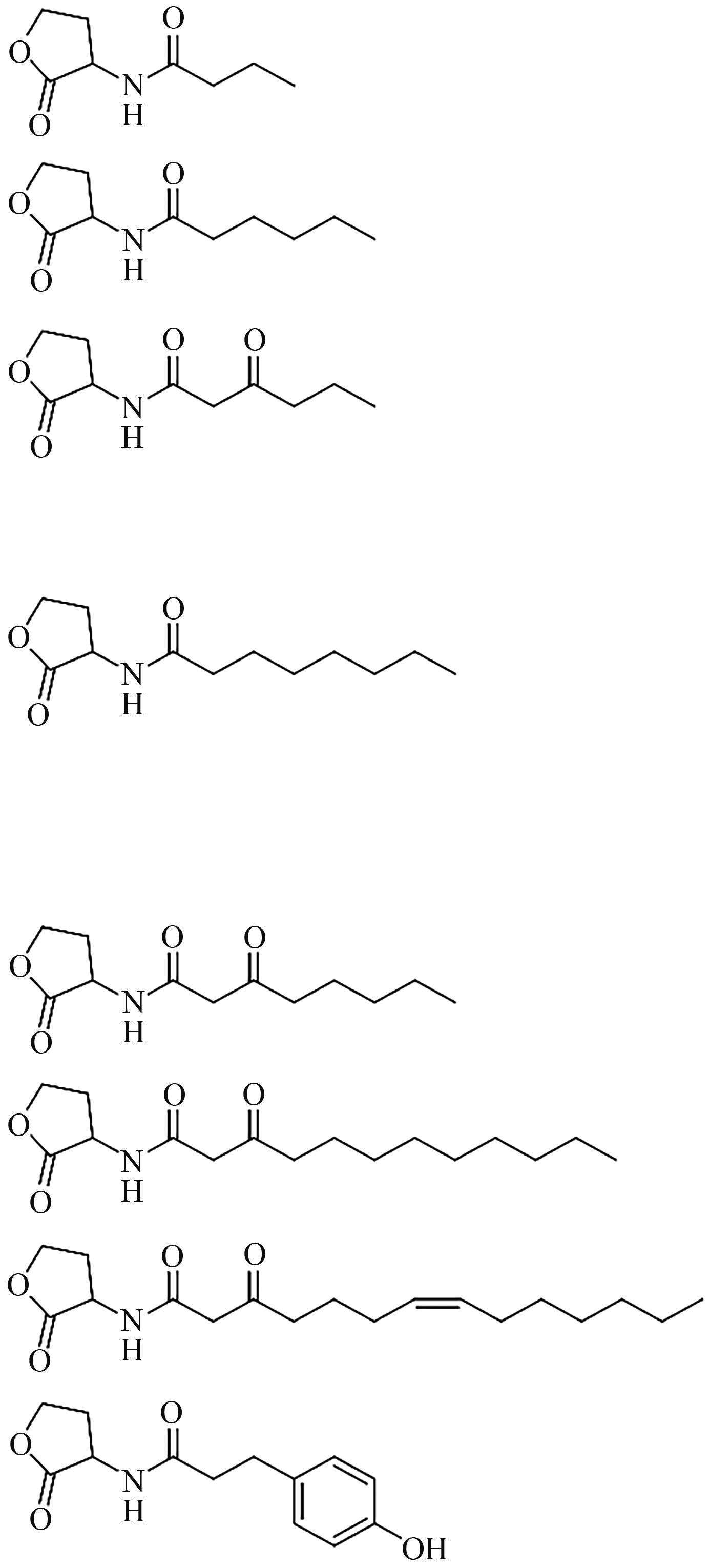 | Pesudomonas aeruginosa |
| Chromobacterium violaceum | ||
| Vibrio fischeri,Pectobacterium atrosepticum,Pectobacterium carotovorum,Pesudomonas syringae | ||
| Urkhoderia cepacian,Agrobacterium tumefaciens,Burkholderia glumae,Gloeothece sp.,Vibrio fischeri | ||
| Agrobacterium tumefaciens,Pectobacterium atrosepticum | ||
| Pesudomonas aeruginosa | ||
| Rhizobium leguminosarum,Vibrio harveyi | ||
| Rhodopesudomonas palustris | ||
| AIs |  | Salmonella typhimurium |
| Vibrio harveyi | ||
| 扩散信号因子(diffusible signal factors,DSFs) |  | Xanthomonas campestris |
| Xylella fastidiosa |
Table 1 Chemical structures of some quorum-sensing signals and molecules present in some microorganisms
| 群体感应信号 | 信号分子结构 | 微生物 |
|---|---|---|
| AHLs |  | Pesudomonas aeruginosa |
| Chromobacterium violaceum | ||
| Vibrio fischeri,Pectobacterium atrosepticum,Pectobacterium carotovorum,Pesudomonas syringae | ||
| Urkhoderia cepacian,Agrobacterium tumefaciens,Burkholderia glumae,Gloeothece sp.,Vibrio fischeri | ||
| Agrobacterium tumefaciens,Pectobacterium atrosepticum | ||
| Pesudomonas aeruginosa | ||
| Rhizobium leguminosarum,Vibrio harveyi | ||
| Rhodopesudomonas palustris | ||
| AIs |  | Salmonella typhimurium |
| Vibrio harveyi | ||
| 扩散信号因子(diffusible signal factors,DSFs) |  | Xanthomonas campestris |
| Xylella fastidiosa |
| [1] | 王博轩,陈佳鑫,蔡佳君,等.海洋链霉菌隐性次级代谢产物基因簇激活的研究进展[J].生物技术进展,2025,15(1):19-26. |
| WANG B X, CHEN J X, CAI J J, et al.. Research progress on metabolites of marine Streptomyces under the strategy of activating silencing genes[J]. Curr. Biotechnol., 2025, 15(1): 19-26. | |
| [2] | 黄艳,胡建伟,朱红惠.粘细菌次生代谢产物中大环类化合物的研究进展[J].生物技术进展,2012,2(4):263-269. |
| HUANG Y, HU J W, ZHU H H. Research progress in macrocyclic compounds of secondary metabolites from myxobacteria[J]. Curr. Biotechnol., 2012, 2(4): 263-269. | |
| [3] | 陈巧莉,黄杰,陈森瑜,等.海洋链霉菌次级代谢产物研究进展[J].生物技术进展,2023,13(6):844-852. |
| CHEN Q L, HUANG J, CHEN S Y, et al.. Research progress on secondary metabolites of marine Streptomyces [J]. Curr. Biotechnol., 2023, 13(6): 844-852. | |
| [4] | BLIN K, SHAW S, AUGUSTIJN H E, et al.. antiSMASH 7.0: new and improved predictions for detection, regulation, chemical structures and visualisation[J]. Nucleic Acids Res., 2023, 51(W1): 46-50 |
| [5] | ROZE L V, CHANDA A, LINZ J E. Compartmentalization and molecular traffic in secondary metabolism: a new understanding of established cellular processes[J]. Fungal Genet. Biol., 2011, 48(1): 35-48. |
| [6] | ZHANG L, FASOYIN O E, MOLNÁR I, et al.. Secondary metabolites from hypocrealean entomopathogenic fungi: novel bioactive compounds[J]. Nat. Prod. Rep., 2020, 37(9): 1181-1206. |
| [7] | PEDRINI N. Molecular interactions between entomopathogenic fungi (Hypocreales) and their insect host: perspectives from stressful cuticle and hemolymph battlefields and the potential of dual RNA sequencing for future studies[J]. Fungal Biol., 2018, 122(6): 538-545. |
| [8] | MACHELEIDT J, MATTERN D J, FISCHER J, et al.. Regulation and role of fungal secondary metabolites[J]. Annu. Rev. Genet., 2016, 50: 371-392. |
| [9] | KELLER N P. Translating biosynthetic gene clusters into fungal armor and weaponry[J]. Nat. Chem. Biol., 2015, 11(9): 671-677. |
| [10] | SCHERLACH K, HERTWECK C. Mediators of mutualistic microbe-microbe interactions[J]. Nat. Prod. Rep., 2018, 35(4): 303-308. |
| [11] | KELLER N P. Fungal secondary metabolism: regulation, function and drug discovery[J]. Nat. Rev. Microbiol., 2019, 17(3): 167-180. |
| [12] | ZEILINGER S, GUPTA V K, DAHMS T E S, et al.. Friends or foes? Emerging insights from fungal interactions with plants[J]. FEMS Microbiol. Rev., 2016, 40(2): 182-207. |
| [13] | MOLNÁR I, GIBSON D M, KRASNOFF S B. Secondary metabolites from entomopathogenic hypocrealean fungi[J]. Nat. Prod. Rep., 2010, 27(9): 1241-1275. |
| [14] | LIU B L, TZENG Y M. Development and applications of destruxins: a review[J]. Biotechnol. Adv., 2012, 30(6): 1242-1254. |
| [15] | RAPPLEYE C A, EISSENBERG L G, GOLDMAN W E. Histoplasma capsulatumα-(1, 3)-glucan blocks innate immune recognition by the β-glucan receptor[J]. Proc. Natl. Acad. Sci. USA, 2007, 104(4): 1366-1370. |
| [16] | AKOUMIANAKI T, KYRMIZI I, VALSECCHI I, et al.. Aspergillus cell wall melanin blocks LC3-associated phagocytosis to promote pathogenicity[J]. Cell Host Microbe, 2016, 19(1): 79-90. |
| [17] | POYNTNER C, MIRASTSCHIJSKI U, STERFLINGER K, et al.. Transcriptome study of an Exophiala dermatitidis PKS1 mutant on an ex vivo skin model: is melanin important for infection?[J/OL]. Front. Microbiol., 2018, 9: 1457[2025-09-27]. . |
| [18] | ZHAO Y, DING J, YUAN W, et al.. Production of a fungal furocoumarin by a polyketide synthase gene cluster confers the chemo-resistance of Neurospora crassa to the predation by fungivorous arthropods[J]. Environ. Microbiol., 2017, 19(10): 3920-3929. |
| [19] | SPRAKER J E, SANCHEZ L M, LOWE T M, et al.. Ralstonia solanacearum lipopeptide induces chlamydospore development in fungi and facilitates bacterial entry into fungal tissues[J]. ISME J., 2016, 10(9): 2317-2330. |
| [20] | FAN Y, LIU X, KEYHANI N O, et al.. Regulatory cascade and biological activity of Beauveria bassiana oosporein that limits bacterial growth after host death[J]. Proc. Natl. Acad. Sci. USA, 2017, 114(9): 1578-1586. |
| [21] | KHALID S, BACCILE J A, SPRAKER J E, et al.. NRPS-derived isoquinolines and lipopetides mediate antagonism between plant pathogenic fungi and bacteria[J]. ACS Chem. Biol., 2018, 13(1): 171-179. |
| [22] | OH D C, POULSEN M, CURRIE C R, et al.. Dentigerumycin: a bacterial mediator of an ant-fungus symbiosis[J]. Nat. Chem. Biol., 2009, 5(6): 391-393. |
| [23] | DHODARY B, SCHILG M, WIRTH R, et al.. Secondary metabolites from Escovopsis weberi and their role in attacking the garden fungus of leaf-cutting ants[J]. Chemistry, 2018, 24(17): 4445-4452. |
| [24] | TOMASZ A, HOTCHKISS R D. Regulation of the transformability of pneumococcal cultures by macromolecular cell products[J]. Proc. Natl. Acad. Sci. USA, 1964, 51(3): 480-487. |
| [25] | NEALSON K H, PLATT T, HASTINGS J W. Cellular control of the synthesis and activity of the bacterial luminescent system[J]. J. Bacteriol., 1970, 104(1): 313-322. |
| [26] | EBERHARD A, BURLINGAME A L, EBERHARD C, et al.. Structural identification of autoinducer of Photobacterium fischeri luciferase[J]. Biochemistry, 1981, 20(9): 2444-2449. |
| [27] | DONG Y H, WANG L Y, ZHANG L H. Quorum-quenching microbial infections: mechanisms and implications[J]. Philos. Trans. R. Soc. Lond. B Biol. Sci., 2007, 362(1483): 1201-1211. |
| [28] | PAPENFORT K, BASSLER B L. Quorum sensing signal-response systems in gram-negative bacteria[J]. Nat. Rev. Microbiol., 2016, 14(9): 576-588. |
| [29] | SCHUSTER M, LOSTROH C P, OGI T, et al.. Identification, timing, and signal specificity of Pseudomonas aeruginosa quorum-controlled genes: a transcriptome analysis[J]. J. Bacteriol., 2003, 185(7): 2066-2079. |
| [30] | WAGNER V E, BUSHNELL D, PASSADOR L, et al.. Microarray analysis of Pseudomonas aeruginosa quorum-sensing regulons: effects of growth phase and environment[J]. J. Bacteriol., 2003, 185(7): 2080-2095. |
| [31] | KATO J Y, FUNA N, WATANABE H, et al.. Biosynthesis of gamma-butyrolactone autoregulators that switch on secondary metabolism and morphological development in Streptomyces [J]. Proc. Natl. Acad. Sci. USA, 2007, 104(7): 2378-2383. |
| [32] | WANG J, WANG W, WANG L, et al.. A novel role of 'pseudo'γ-butyrolactone receptors in controlling γ-butyrolactone biosynthesis in Streptomyces [J]. Mol. Microbiol., 2011, 82(1): 236-250. |
| [33] | LEE J, WU J, DENG Y, et al.. A cell-cell communication signal integrates quorum sensing and stress response[J]. Nat. Chem. Biol., 2013, 9(5): 339-343. |
| [34] | CHEN H, FUJITA M, FENG Q, et al.. Tyrosol is a quorum-sensing molecule in Candida albicans [J]. Proc. Natl. Acad. Sci. USA, 2004, 101(14): 5048-5052. |
| [35] | CRUZ J M, DOMÍNGUEZ J M, DOMÍNGUEZ H, et al.. Dimorphic behaviour of Debaryomyces hansenii grown on barley bran acid hydrolyzates[J]. Biotechnol. Lett., 2000, 22(7): 605-610. |
| [36] | GORI K, MORTENSEN H D, ARNEBORG N, et al.. Ammonia production and its possible role as a mediator of communication for Debaryomyces hansenii and other cheese-relevant yeast species[J]. J. Dairy Sci., 2007, 90(11): 5032-5041. |
| [37] | HOMER C M, SUMMERS D K, GORANOV A I, et al.. Intracellular action of a secreted peptide required for fungal virulence[J]. Cell Host Microbe, 2016, 19(6): 849-864. |
| [38] | TIAN X, HE G J, HU P, et al.. Cryptococcus neoformans sexual reproduction is controlled by a quorum sensing peptide[J]. Nat. Microbiol., 2018, 3(6): 698-707. |
| [39] | RAINA S, ODELL M, KESHAVARZ T. Quorum sensing as a method for improving sclerotiorin production in Penicillium sclerotiorum [J]. J. Biotechnol., 2010, 148(2-3): 91-98. |
| [40] | JI G, BEAVIS R C, NOVICK R P. Cell density control of staphylococcal virulence mediated by an octapeptide pheromone[J]. Proc. Natl. Acad. Sci. USA, 1995, 92(26): 12055-12059. |
| [41] | MAYVILLE P, JI G, BEAVIS R, et al.. Structure-activity analysis of synthetic autoinducing thiolactone peptides from Staphylococcus aureus responsible for virulence[J]. Proc. Natl. Acad. Sci. USA, 1999, 96(4): 1218-1223. |
| [42] | TAKAHASHI M, IWASAKI S, KOBAYASHI H, et al.. Studies on macrocyclic lactone antibiotics. XI. anti-mitotic and anti-tubulin activity of new antitumor antibiotics, rhizoxin and its homologues[J]. J. Antibiot., 1987, 40(1): 66-72. |
| [43] | LINIGER M, NEUHAUS C M, ALTMANN K H. Ring-closing metathesis approaches towards the total synthesis of rhizoxins[J/OL]. Molecules, 2020, 25 (19): 4527[2025-09-12]. . |
| [44] | PARTIDA-MARTINEZ L P, HERTWECK C. Pathogenic fungus harbours endosymbiotic bacteria for toxin production[J]. Nature, 2005, 437(7060): 884-888. |
| [45] | SCHERLACH K, BUSCH B, LACKNER G, et al.. Symbiotic cooperation in the biosynthesis of a phytotoxin[J]. Angew. Chem. Int. Ed., 2012, 51(38): 9615-9618. |
| [46] | PARTIDA-MARTINEZ L P, DE LOOSS C F, ISHIDA K, et al.. Rhizonin, the first mycotoxin isolated from the zygomycota, is not a fungal metabolite but is produced by bacterial endosymbionts[J]. Appl. Environ. Microbiol., 2007, 73(3): 793-797. |
| [47] | FRASES S, CHASKES S, DADACHOVA E, et al.. Induction by Klebsiella aerogenes of a melanin-like pigment in Cryptococcus neoformans [J]. Appl. Environ. Microbiol., 2006, 72(2): 1542-1550. |
| [48] | CALVO A M, GARDNER H W, KELLER N P. Genetic connection between fatty acid metabolism and sporulation in Aspergillus nidulans [J]. J. Biol. Chem., 2001, 276(28): 25766-25774. |
| [49] | ZHENG H, KIM J, LIEW M, et al.. Redox metabolites signal polymicrobial biofilm development via the NapA oxidative stress cascade in Aspergillus [J]. Curr. Biol., 2015, 25(1): 29-37. |
| [50] | STUDT L, WIEMANN P, KLEIGREWE K, et al.. Biosynthesis of fusarubins accounts for pigmentation of Fusarium fujikuroi perithecia[J]. Appl. Environ. Microbiol., 2012, 78(12): 4468-4480. |
| [51] | SCHINDLER D, NOWROUSIAN M. The polyketide synthase gene pks4 is essential for sexual development and regulates fruiting body morphology in Sordaria macrospora [J]. Fungal Genet. Biol., 2014, 68: 48-59. |
| [52] | LEONARD K J. Virulence, temperature optima, and competitive abilities of isolines of races T and O of Bipolaris maydis [J/OL]. Phytopathology, 1977, 77(10): 1273[2025-09-27]. . |
| [53] | EISENMAN H C, CASADEVALL A. Synthesis and assembly of fungal melanin[J]. Appl. Microbiol. Biotechnol., 2012, 93(3): 931-940. |
| [54] | SHUKLA S, PARK J, KIM D H, et al.. Total phenolic content, antioxidant, tyrosinase and α-glucosidase inhibitory activities of water soluble extracts of noble starter culture Doenjang, a Korean fermented soybean sauce variety[J]. Food Control, 2016, 59: 854-861. |
| [55] | SINGARAVELAN N, GRISHKAN I, BEHARAV A, et al.. Adaptive melanin response of the soil fungus Aspergillus niger to UV radiation stress at "Evolution Canyon", Mount Carmel, Israel[J/OL]. PloS ONE, 2008, 3 (8): e2993[2025-09-12]. |
| [56] | JACOBSON E S. Pathogenic roles for fungal melanins[J]. Clin. Microbiol. Rev., 2000, 13(4): 708-717. |
| [57] | ZHAO L, KIM J C, PAIK M J, et al.. A multifunctional and possible skin UV protectant, (3R)-5-hydroxymellein, produced by an endolichenic fungus isolated from Parmotrema austrosinense [J/OL]. Molecules, 2016, 22 (1): 26[2025-09-12]. . |
| [58] | 潘少婷,王博轩,陈佳鑫,等.海洋真菌来源的聚酮类化合物研究进展[J].生物技术进展,2024,14(6):993-1003. |
| PAN S T, WANG B X, CHEN J X, et al.. Research progress on polyketides from marine fungi[J]. Curr. Biotechnol., 2024, 14(6): 993-1003. | |
| [59] | YANG X, CHEN Y, ZHANG L, et al.. Melanin precursors mediated adaption to temperature changes in fungus and animal via inhibition of lipid-mediated ferroptosis[J]. Sci. China Life Sci., 2023, 66(8): 1800-1817. |
| [60] | LI S, WANG D, HE J, et al.. Thermophilic fungus uses anthraquinones to modulate ferrous excretion, sterol-mediated endocytosis, and iron storage in response to cold stress[J/OL]. Microb. Biotechnol., 2024, 17 (9): e70002[2025-09-12]. . |
| [1] | Mingjie LYU, Guiying JIA, Xinran LI, Yaning GUO, Kai LI, Rui CHEN, Liang LI. Research Progress on Crop Substantive Derived Variety System and Judgment Method [J]. Current Biotechnology, 2025, 15(5): 764-772. |
| [2] | Tianyuan ZHAO, Jing WANG, Yulu WANG, Chunsen YUAN, Xuechai CHEN. Research Progress on Neuroprotective Effects of Betaine [J]. Current Biotechnology, 2025, 15(2): 220-225. |
| [3] | Xiaoqi WU, Wenjing GONG, Guoyu LI, Ang LI, Jihua WANG, Di CUI. Knowledge Gaps and Chanllenges in Microbial Fermentation of Traditional Chinese Medicine: From Strain Selection to Quality Control [J]. Current Biotechnology, 2025, 15(2): 201-211. |
| [4] | Sujie WANG, Mengli GU, Jiemeng TAO, Zhijun TONG, Junjia GUO, Jingjing JIN, Mengxiao XU, Lijun MENG, Jianfeng ZHANG, Peijian CAO, Peng LU. Isolation, Identification and Whole-genome Sequence Analysis of Phytophthora nicotianae Antagonistic Bacteria XC-29 [J]. Current Biotechnology, 2024, 14(6): 1004-1015. |
| [5] | Huanzhen WU, Ye YANG, Xiuming CUI, Yuan LIU. The Current Status and Improvement Strategies of Agricultural Biological Control Technology [J]. Current Biotechnology, 2024, 14(5): 697-711. |
| [6] | Bicong WU, Bo JIAO, Yu ZHANG, Xin GUO, Yu ZHANG, Xiaohong LUO, Lei DAI, Qiang WANG. Effect of Feed-to-liquid Ratio on the Quality Characteristics of Stirred UHT Walnut Yogurt [J]. Current Biotechnology, 2024, 14(4): 640-648. |
| [7] | Kai ZOU. Research and Application of Plant Hairy Root [J]. Current Biotechnology, 2024, 14(3): 341-348. |
| [8] | Liwen WANG, Jiangkun WANG, Bingbing WANG, Jianhong XU, Jianrong SHI, Xin LIU. Roles of Fusarium Toxins in Plant-pathogen Interaction [J]. Current Biotechnology, 2024, 14(2): 182-188. |
| [9] | Qiaoli CHEN, Jie HUANG, Senyu CHEN, Shaoting PAN, Lingzhi TANG, Xuan HONG. Research Progress on Secondary Metabolites of Marine Streptomyces [J]. Current Biotechnology, 2023, 13(6): 844-852. |
| [10] | Kainan SONG, Linan XIE, Yuquan XU. Progress of Fungal Secondary Metabolites with Potential Herbicidal Activity [J]. Current Biotechnology, 2023, 13(2): 181-194. |
| [11] | Yang YANG, Fenglin WANG, De LIU, Yuanyuan LUO, Jianhua ZHU. Research Progress of CRISPR⁃Cas9 Technology on the Production of Plant Secondary Metabolites [J]. Current Biotechnology, 2022, 12(6): 806-816. |
| [12] | Xin LIU, Xin FANG, Shuang WANG, Liwen WANG, Deliang WU, Yin Won LEE, Sherif Ramzy MOHAMED, Jianhong XU, Jianrong SHI. Subcellular Localization and Cellular Machinery Required for Deoxynivalenol Assembly: Updates and New Insights [J]. Current Biotechnology, 2021, 11(5): 642-646. |
| [13] | WU Chunlei1, WU Zhilin2, WU Jie2, JIANG Han2, ZANG Huawei2, TAN Genjia2, YUAN Linxi3, LI Miao2*. Progress on Application of Selenium in Plant Protection and Agricultural Products Quality and Safety [J]. Curr. Biotech., 2017, 7(5): 462-466. |
| [14] | GHUNIQIGUL Abake, AYNUR Yusup, MAYNUR Dawut, QIMENGUL Turghun, YIMIT Rahman*. Progress on Pharmacological Effects of Flavonoids from Propolis [J]. Curr. Biotech., 2017, 7(1): 13-19. |
| [15] | LI Gui-zhen1,2, LAI Qi-liang2, YAN Pei-sheng1,3*, SHAO Zong-ze1,2*. Advance on Marine Petroleum Pollution and Microbial Remediation [J]. Curr. Biotech., 2015, 5(3): 164-169. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||