


生物技术进展 ›› 2026, Vol. 16 ›› Issue (2): 309-317.DOI: 10.19586/j.2095-2341.2025.0126
• 进展评述 • 上一篇
王育萌( ), 赵鹏翔(
), 赵鹏翔( ), 张旭娟, 党政, 李函, 刘子怡, 王佳腾, 于思雪, 马雪梅(
), 张旭娟, 党政, 李函, 刘子怡, 王佳腾, 于思雪, 马雪梅( )
)
收稿日期:2025-09-23
接受日期:2025-12-17
出版日期:2026-03-25
发布日期:2026-04-27
通讯作者:
赵鹏翔,马雪梅
作者简介:王育萌E-mail: wangyumeng@bjut.edu.cn
基金资助:
Yumeng WANG( ), Pengxiang ZHAO(
), Pengxiang ZHAO( ), Xujuan ZHANG, Zheng DANG, Han LI, Ziyi LIU, Jiateng WANG, Sixue YU, Xuemei MA(
), Xujuan ZHANG, Zheng DANG, Han LI, Ziyi LIU, Jiateng WANG, Sixue YU, Xuemei MA( )
)
Received:2025-09-23
Accepted:2025-12-17
Online:2026-03-25
Published:2026-04-27
Contact:
Pengxiang ZHAO,Xuemei MA
摘要:
肝脏是人体代谢和解毒的核心器官,其功能维持依赖于复杂的酶系统网络,其中细胞色素P450(cytochrome P450,CYP450)酶系通过催化药物Ⅰ相代谢参与外源性物质的解毒或毒性转化,其活性调控对药物疗效及用药安全具有重要影响。CYP450在肝脏疾病、心血管、肿瘤等多种疾病的发生发展中具有重要的治疗和病理生理学意义,深入研究CYP450的功能及其在疾病中的变化,可为开发新的治疗策略提供重要依据。重点阐述了CYP450抑制剂和诱导剂在临床中的应用,总结了靶向调控CYP450对优化药物代谢提升疗效、降低不良反应及推动个体化用药的重要意义,以期为肝脏疾病精准治疗及拓展应用提供新方向。
中图分类号:
王育萌, 赵鹏翔, 张旭娟, 党政, 李函, 刘子怡, 王佳腾, 于思雪, 马雪梅. 细胞色素P450酶系药物研发:肝脏药物代谢调控与临床应用研究进展[J]. 生物技术进展, 2026, 16(2): 309-317.
Yumeng WANG, Pengxiang ZHAO, Xujuan ZHANG, Zheng DANG, Han LI, Ziyi LIU, Jiateng WANG, Sixue YU, Xuemei MA. Cytochrome P450 Enzyme System in Drug Development: Advances in Research on Regulation of Hepatic Drug Metabolism and Clinical Applications[J]. Current Biotechnology, 2026, 16(2): 309-317.
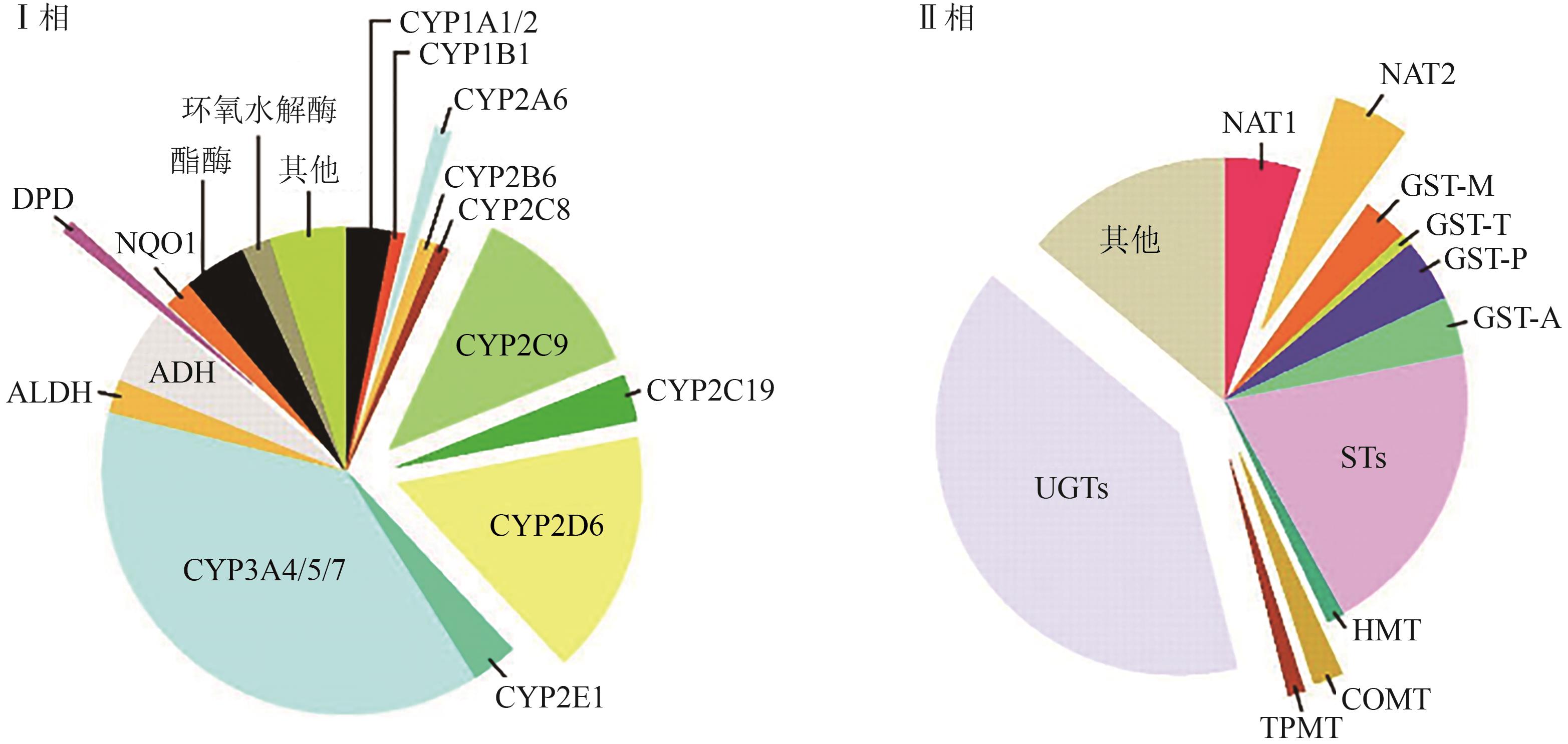
图2 人体药物代谢Ⅰ相Ⅱ相多态性代谢酶[11]注:该图体现药物代谢贡献的大致结果,酶对应与药物效应个体差异相关的多态性代谢酶。ADH—酒精脱氢酶;ALDH—醛脱氢酶;CYP—细胞色素P450;DPD—二氢嘧啶脱氢酶;NQO1—醌氧化还原酶;NADPH—还原型烟酰胺腺嘌呤二核苷酸磷酸;COMT—儿茶酚O-甲基转移酶;GST—谷胱甘肽S-转移酶;HMT—组胺甲基转移酶;NAT—N-乙酰转移酶;STs—磺基转移酶;TPMT—硫嘌呤甲基转移酶;UGTs—尿苷5'-三磷酸葡萄糖醛酸转移酶。
Fig. 2 Phase Ⅰ/Ⅱ drug metabolism in humans and drug-metabolizing enzymes[11]
| 药物类型 | 药物名称 | 主要代谢酶 | 临床应用/风险提示 | 参考文献 |
|---|---|---|---|---|
| 精神类药物 | 奥氮平 | CYP1A2、CYP2D6 | 测定患者白细胞中CYP1A2 mRNA 水平,以评估奥氮平药效 | [ |
| 利培酮 | CYP2D6 | CYP2D6多态性显著影响利培酮的血浆浓度,导致不良反应 | [ | |
| 心血管药 | 辛伐他汀 | CYP3A4、CYP2A6 | 避免CYP3A4的强抑制剂和他汀类药物共同给药导致的横纹肌溶解症 | [ |
| 氯吡格雷 | CYP2C19 | CYP2C19功能丧失会导致氯吡格雷激活受阻、血小板聚集减少较小以及治疗失败高风险 | [ | |
| 抗疟疾药 | 青蒿素 | CYP2B6 | 青蒿甲醇提取物对CYP2B6在体外具有不可逆的抑制作用 | [ |
| 抗凝药 | 华法林 | CYP2C9 | 携带CYP2C9变异基因的患者需降低华法林剂量,防止出血风险显著增加;可通过基因检测预测最佳起始剂量 | [ |
| 抗炎药 | 布洛芬 | CYP2C9 | 代谢受CYP2C9多态性影响,可能增加胃肠道出血风险 | [ |
| 双氯芬酸 | CYP2C9 | |||
| 止痛解热药 | 对乙酰氨基酚 | CYP2E1 | 降低CYP2E1活性可以减少对乙酰氨基酚代谢过程中有毒中间代谢物的产生 | [ |
| 抗菌药 | 利福平 | CYP3A4 | 可引起环孢菌素等免疫抑制药物治疗患者出现急性移植排斥反应 | [ |
| 抗肿瘤药 | 紫杉醇 | CYP3A4、CYP2C8 | 紫杉醇代谢的个体差异会导致毒性增加,需要药物检测合计个性化剂量以提高总体疗效 | [ |
| 他莫昔芬 | CYP2D6 | 饮食标志物茄碱可作为体内表型标志物快速识别CYP2D6功能低下者,适用于基因检测结果未出或已用CYP2D6药物干扰情况 | [ |
表1 CYP450家族参与代谢的部分常见药物
Table 1 Common drugs metabolized by the CYP450 family
| 药物类型 | 药物名称 | 主要代谢酶 | 临床应用/风险提示 | 参考文献 |
|---|---|---|---|---|
| 精神类药物 | 奥氮平 | CYP1A2、CYP2D6 | 测定患者白细胞中CYP1A2 mRNA 水平,以评估奥氮平药效 | [ |
| 利培酮 | CYP2D6 | CYP2D6多态性显著影响利培酮的血浆浓度,导致不良反应 | [ | |
| 心血管药 | 辛伐他汀 | CYP3A4、CYP2A6 | 避免CYP3A4的强抑制剂和他汀类药物共同给药导致的横纹肌溶解症 | [ |
| 氯吡格雷 | CYP2C19 | CYP2C19功能丧失会导致氯吡格雷激活受阻、血小板聚集减少较小以及治疗失败高风险 | [ | |
| 抗疟疾药 | 青蒿素 | CYP2B6 | 青蒿甲醇提取物对CYP2B6在体外具有不可逆的抑制作用 | [ |
| 抗凝药 | 华法林 | CYP2C9 | 携带CYP2C9变异基因的患者需降低华法林剂量,防止出血风险显著增加;可通过基因检测预测最佳起始剂量 | [ |
| 抗炎药 | 布洛芬 | CYP2C9 | 代谢受CYP2C9多态性影响,可能增加胃肠道出血风险 | [ |
| 双氯芬酸 | CYP2C9 | |||
| 止痛解热药 | 对乙酰氨基酚 | CYP2E1 | 降低CYP2E1活性可以减少对乙酰氨基酚代谢过程中有毒中间代谢物的产生 | [ |
| 抗菌药 | 利福平 | CYP3A4 | 可引起环孢菌素等免疫抑制药物治疗患者出现急性移植排斥反应 | [ |
| 抗肿瘤药 | 紫杉醇 | CYP3A4、CYP2C8 | 紫杉醇代谢的个体差异会导致毒性增加,需要药物检测合计个性化剂量以提高总体疗效 | [ |
| 他莫昔芬 | CYP2D6 | 饮食标志物茄碱可作为体内表型标志物快速识别CYP2D6功能低下者,适用于基因检测结果未出或已用CYP2D6药物干扰情况 | [ |
| 名称 | CYP450类型 | 临床效果 | 参考文献 | |
|---|---|---|---|---|
| 抑制剂 | 酮康唑 | CYP3A4 | 高选择性广谱抗真菌 | [ |
| COR-500015 | CYP17 | 调节皮质醇的产生 | [ | |
| 氟康唑 | CYP2C9 | 降低交感神经血管收缩 | [ | |
| 新型异恶唑衍生物 | CYP1A2 | - | [ | |
| 类黄酮 | CYP3A4 | 高剂量使用引发不良反应 | [ | |
| 化合物9 | CYP4Z1 | 有效抑制乳腺癌细胞 | [ | |
| 黄精多糖 | CYP2C9/2D6/3A4 | 草药共同给药时避免DDI | [ | |
| 光敏Ru(Ⅱ)配合物 | CYP3A4 | 增强抗癌药物的治疗效果 | [ | |
| 噻唑酰胺化合物B20 | CYP1B1 | 靶向治疗克服紫杉醇耐药性 | [ | |
| 胡椒碱 | CYP2J2 | 抗肿瘤,降低线粒体功能障碍 | [ | |
| 紫苏醇 | CYP2A6/CYP2B6 | - | [ | |
| 诱导剂 | 聚乙二醇化脂质体 | CYP3A | 增强聚乙二醇抗肝细胞癌作用 | [ |
| 诃子 | CYP1A2等 | 显著改善肝功能和组织病理学 | [ | |
| 厄洛替尼 | CYPA3 | 治疗晚期非小细胞肺癌 | [ | |
| 黄芪注射液 | CYP2D1 | 提高癌症疗效,降低耐药性 | [ |
表2 新型CYP450抑制剂和诱导剂总结
Table 2 Summary of novel CYP450 inhibitors and inducers
| 名称 | CYP450类型 | 临床效果 | 参考文献 | |
|---|---|---|---|---|
| 抑制剂 | 酮康唑 | CYP3A4 | 高选择性广谱抗真菌 | [ |
| COR-500015 | CYP17 | 调节皮质醇的产生 | [ | |
| 氟康唑 | CYP2C9 | 降低交感神经血管收缩 | [ | |
| 新型异恶唑衍生物 | CYP1A2 | - | [ | |
| 类黄酮 | CYP3A4 | 高剂量使用引发不良反应 | [ | |
| 化合物9 | CYP4Z1 | 有效抑制乳腺癌细胞 | [ | |
| 黄精多糖 | CYP2C9/2D6/3A4 | 草药共同给药时避免DDI | [ | |
| 光敏Ru(Ⅱ)配合物 | CYP3A4 | 增强抗癌药物的治疗效果 | [ | |
| 噻唑酰胺化合物B20 | CYP1B1 | 靶向治疗克服紫杉醇耐药性 | [ | |
| 胡椒碱 | CYP2J2 | 抗肿瘤,降低线粒体功能障碍 | [ | |
| 紫苏醇 | CYP2A6/CYP2B6 | - | [ | |
| 诱导剂 | 聚乙二醇化脂质体 | CYP3A | 增强聚乙二醇抗肝细胞癌作用 | [ |
| 诃子 | CYP1A2等 | 显著改善肝功能和组织病理学 | [ | |
| 厄洛替尼 | CYPA3 | 治疗晚期非小细胞肺癌 | [ | |
| 黄芪注射液 | CYP2D1 | 提高癌症疗效,降低耐药性 | [ |
| [1] | KOSICKA-NOWORZYŃ K, ROMANIUK-DRAPAŁA A, SHENG Y H, et al.. Obesity-related drug-metabolizing enzyme expression alterations in the human liver[J/OL]. Biomed. Pharmacother., 2025, 187: 118155[2026-01-11]. . |
| [2] | ZHAO Y, WU Z, LI J, et al.. The key role of cytochrome P450s in the biosynthesis of plant derived natural products[J/OL]. Plant Physiol. Biochem., 2025, 222: 1096955[2026-01-11]. . |
| [3] | RAO GAJULA S N, PILLAI M S, SAMANTHULA G, et al.. Cytochrome P450 enzymes: a review on drug metabolizing enzyme inhibition studies in drug discovery and development[J]. Bioanalysis, 2021, 13(17): 1355-1378. |
| [4] | GUENGERICH F P. Cytochrome P450 enzymes as drug targets in human disease[J]. Drug Metab. Dispos., 2024, 52(6): 493-497. |
| [5] | ZHANG Y, RUGGIERO M, HAGENBUCH B. OATP1B3 expression and function is modulated by coexpression with OCT1, OATP1B1, and NTCP[J]. Drug Metab. Dispos., 2020, 48(8): 622-630. |
| [6] | LI T, FENG Y, LIU Y, et al.. The role of organic anion transport peptides in cyclophosphamide-induced hepatotoxicity in high-fat diet mice[J/OL]. Life Sci., 2024, 359: 1232395[2026-01-11]. . |
| [7] | JAMWAL R, BARLOCK B J. Nonalcoholic fatty liver disease (NAFLD) and hepatic cytochrome P450 (CYP) enzymes[J/OL]. Pharmaceuticals, 2020, 13(9): 222[2026-01-11]. . |
| [8] | STANLEY L A. Chapter 26 - Drug metabolism [M]//MCCREATH S B, CLEMENT Y N. Pharmacognosy (Second Edition). Academic Press. 2024: 597-624. |
| [9] | WANG Y, LIU Y, YAO H, et al.. Decreased plasma exposure of clopidogrel active metabolite in rats after long-term treatment with clopidogrel[J]. Biopharm. Drug Dispos., 2023, 44(2): 129-136. |
| [10] | FUKAMI T, YOKOI T, NAKAJIMA M. Non-P450 drug-metabolizing enzymes: contribution to drug disposition, toxicity, and development[J]. Annu. Rev. Pharmacol. Toxicol., 2022, 62: 405-425. |
| [11] | EVANS W E, RELLING M V. Pharmacogenomics: translating functional genomics into rational therapeutics[J]. Science, 1999, 286(5439): 487-491. |
| [12] | WANG Y, CHEN Q, WU S, et al.. Amelioration of ethanol-induced oxidative stress and alcoholic liver disease by in vivo RNAi targeting Cyp2e1[J]. Acta Pharm. Sin. B, 2023, 13(9): 3906-3918. |
| [13] | CORSINI A, BORTOLINI M. Drug-induced liver injury: the role of drug metabolism and transport[J]. J. Clin. Pharmacol., 2013, 53(5): 463-474. |
| [14] | WARING R H. Cytochrome P450: genotype to phenotype[J]. Xenobiotica, 2020, 50(1): 9-18. |
| [15] | FEKETE F, MENUS Á, TÓTH K, et al.. CYP 1A2 expression rather than genotype is associated with olanzapine concentration in psychiatric patients[J/OL]. Sci. Rep., 2023, 13: 18507[2026-01-11]. . |
| [16] | DE BRABANDER E, KLEINE SCHAARS K, VAN AMELSVOORT T, et al.. Influence of CYP2C19 and CYP2D6 on side effects of aripiprazole and risperidone: a systematic review[J]. J. Psychiatr. Res., 2024, 174: 137-152. |
| [17] | HOUGAARD CHRISTENSEN M M, BRUUN HAASTRUP M, ØHLENSCHLAEGER T, et al.. Interaction potential between clarithromycin and individual statins-a systematic review[J]. Basic Clin. Pharmacol. Toxicol., 2020, 126(4): 307-317. |
| [18] | PATEL R C, THOMAS C D, ROSSI J S, et al.. CYP 2C19 phenotype, P2Y(12) inhibitor selection, and clinical outcomes in patients on maintenance clopidogrel therapy[J/OL]. J. Am. Heart Assoc., 2025, 14(14): e041634[2026-01-11]. . |
| [19] | KONDŽA M, MANDIĆ M, IVANČIĆ I, et al.. Artemisia annua L. extracts irreversibly inhibit the activity of CYP2B6 and CYP3A4 enzymes[J/OL]. Biomedicines, 2023, 11(1): 232[2026-01-11]. . |
| [20] | LINDLEY K J, LIMDI N A, CAVALLARI L H, et al.. Warfarin dosing in patients with CYP2C9*5 variant alleles[J]. Clin. Pharmacol. Ther., 2022, 111(4): 950-955. |
| [21] | MACÍAS Y, GÓMEZ TABALES J, GARCÍA-MARTÍN E, et al.. An update on the pharmacogenomics of NSAID metabolism and the risk of gastrointestinal bleeding[J]. Expert Opin. Drug Metab. Toxicol., 2020, 16(4): 319-332. |
| [22] | ZHANG L, ZHANG F, XIAO Y, et al.. The nuclear receptor REV-ERBα regulates CYP2E1 expression and acetaminophen hepatotoxicity[J]. Xenobiotica, 2022, 52(6): 633-643. |
| [23] | NIEMI M, BACKMAN J T, FROMM M F, et al.. Pharmacokinetic interactions with rifampicin: clinical relevance[J]. Clin. Pharmacokinet., 2003, 42(9): 819-850. |
| [24] | RYU J H, YU J, JEON J S, et al.. Heterotropic activation of cytochrome P450 3A4 by perillyl alcohol[J/OL]. Pharmaceutics, 2024, 16(12): 1581[2026-01-11]. . |
| [25] | KUMAR V, VARDHAN G, SEHRAWAT A, et al.. A novel ultra-performance liquid chromatography detection method development and validation for paclitaxel and its major metabolite in human plasma[J]. Indian J. Pharmacol., 2024, 56(4): 253-259. |
| [26] | MEDWID S, SCHWARZ U I, CHOI Y H, et al.. Solanidine metabolites as diet-derived biomarkers of CYP2D6-mediated tamoxifen metabolism in breast cancer patients[J]. Clin. Pharmacol. Ther., 2024, 116(5): 1269-1277. |
| [27] | HE J, LIU X, LI C. Engineering electron transfer pathway of cytochrome P 450s[J/OL]. Molecules, 2024, 29(11): 2480[2026-01-11]. |
| [28] | NEBERT D W, WIKVALL K, MILLER W L. Human cytochromes P450 in health and disease[J/OL]. Philos. Trans. R. Soc. Lond. B Biol. Sci., 2013, 368(1612): 20120431[2026-01-11]. . |
| [29] | ZHU Q, XIE X, FANG L, et al.. Chronic alcohol intake disrupts cytochrome P450 enzyme activity in alcoholic fatty liver disease: insights into metabolic alterations and therapeutic targets[J/OL]. Front. Chem., 2025, 13: 1509785[2026-01-11]. . |
| [30] | GETHINGS L A, GRAY N, PLUMB R S, et al.. Proteomic consequences of the deletion of cytochrome P450 (CYP450) reductase in mice[J/OL]. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci., 2021, 1179: 122803[2026-01-11]. . |
| [31] | JIANG Y J, CAO Y M, CAO Y B, et al.. A review: cytochrome P450 in alcoholic and non-alcoholic fatty liver disease[J]. Diabetes Metab. Syndr. Obes., 2024, 17: 1511-1521. |
| [32] | ELBEKAI R H, KORASHY H M, EL-KADI A O S. The effect of liver cirrhosis on the regulation and expression of drug metabolizing enzymes[J]. Curr. Drug Metab., 2004, 5(2): 157-167. |
| [33] | MAST N, ANDERSON K W, JOHNSON K M, et al.. In vitro cytochrome P450 46A1 (CYP46A1) activation by neuroactive compounds[J]. J. Biol. Chem., 2017, 292(31): 12934-12946. |
| [34] | GAO J, WANG Z, WANG G J, et al.. From hepatofibrosis to hepatocarcinogenesis: higher cytochrome P450 2E1 activity is a potential risk factor[J]. Mol. Carcinog., 2018, 57(10): 1371-1382. |
| [35] | CHI X, ZHANG H, WU H, et al.. Discovery of novel tetrazoles featuring a pyrazole moiety as potent and highly selective antifungal agents[J]. ACS Omega, 2023, 8(19): 17103-17115. |
| [36] | SCHROEDER R L, TRAM P, LIU J, et al.. Novel functionalized 5-(phenoxymethyl)-1, 3-dioxane analogs exhibiting cytochrome P450 inhibition: a patent evaluation WO2015048311 (A1)[J]. Expert Opin. Ther. Pat., 2016, 26(1): 139-147. |
| [37] | BUELOW A A, MATNEY J E, SKILLETT S M, et al.. Inhibition of CYP450 pathways reduces functional sympatholysis in healthy young adults[J]. Am. J. Physiol. Regul. Integr. Comp. Physiol., 2025, 328(6): 642-650. |
| [38] | WAZALWAR S S, BANPURKAR A R, PERDIH F. Synthesis, crystal structure and molecular docking study of novel isoxazole derivatives as CYP450 inhibitors in search of anticancer agents[J]. J. Biomol. Struct. Dyn., 2023, 41(19): 9476-9491. |
| [39] | KONDŽA M, BRIZIĆ I, JOKIĆ S. Flavonoids as CYP 3A4 inhibitors in vitro[J/OL]. Biomedicines, 2024, 12(3): 644[2026-01-11]. . |
| [40] | MACHALZ D, LI H, DU W, et al.. Discovery of a novel potent cytochrome P450 CYP4Z1 inhibitor[J/OL]. Eur. J. Med. Chem., 2021, 215: 113255[2026-01-11]. . |
| [41] | DUAN Y, WANG X, WANG R, et al.. Inhibitory effects characteristics of polysaccharide of Polygonati rhizome on cytochrome P450 enzymes[J]. Naunyn Schmiedebergs Arch. Pharmacol., 2025, 398(4): 4087-4094. |
| [42] | TOUPIN N, STEINKE S J, NADELLA S, et al.. Photosensitive Ru(Ⅱ) complexes as inhibitors of the major human drug metabolizing enzyme CYP3A4[J]. J. Am. Chem. Soc., 2021, 143(24): 9191-9205. |
| [43] | WANG T, ZHANG H, TAN D, et al.. Discovery of highly selective CYP1B1 inhibitors[J]. J. Med. Chem., 2025, 68(12): 13089-13112. |
| [44] | TIAN X, ZHOU M, NING J, et al.. The development of novel cytochrome P450 2J2 (CYP2J2) inhibitor and the underlying interaction between inhibitor and CYP2J2[J]. J. Enzyme Inhib. Med. Chem., 2021, 36(1): 737-748. |
| [45] | ZHANG X, PAN J, YE X, et al.. Activation of CYP3A by accelerated blood clearance phenomenon potentiates the hepatocellular carcinoma-targeting therapeutic effects of PEGylated anticancer prodrug liposomes[J]. Drug Metab. Dispos., 2023, 51(12): 1651-1662. |
| [46] | ZHU J, AN M, WANG W, et al.. Potential induction of the relative mRNA expression levels of CYP 450 by Zhicaowu-Hezi (Aconiti kusnezoffii Radix preparata and Terminalia chebula Retz.)[J/OL]. Front. Pharmacol., 2025, 16: 1573739[2026-01-11]. . |
| [47] | SVEDBERG A, VIKINGSSON S, VIKSTRÖM A, et al.. Erlotinib treatment induces cytochrome P450 3A activity in non-small cell lung cancer patients[J]. Br. J. Clin. Pharmacol., 2019, 85(8): 1704-1709. |
| [48] | SHI W, LIU T, WANG K, et al.. Astragalus Injection modulates the pharmacokinetics of doxorubicin and CYP450 enzymes[J]. Curr. Pharm. Des., 2025, 31(40): 3234-3246. |
| [49] | TAN B H, AHEMAD N, PAN Y, et al.. Mechanism-based inactivation of cytochromes P450: implications in drug interactions and pharmacotherapy[J]. Xenobiotica, 2024, 54(9): 575-598. |
| [50] | QIU J, LU J, WANG X, et al.. Discovery of a CYP 2E1 inhibitor and its therapeutic potential in severe acute pancreatitis[J/OL]. Eur. J. Med. Chem., 2025, 292: 117666[2026-01-11]. . |
| [51] | WÓJCIKOWSKI J, DANEK P J, BASIŃSKA-ZIOBROŃ A, et al.. In vitro inhibition of human cytochrome P450 enzymes by the novel atypical antipsychotic drug asenapine: a prediction of possible drug-drug interactions[J]. Pharmacol. Rep., 2020, 72(3): 612-621. |
| [52] | ZHAO L, SUN N, TIAN L, et al.. Strategies for the development of highly selective cytochrome P450 inhibitors: several CYP targets in current research[J]. Bioorg. Med. Chem. Lett., 2019, 29(16): 2016-2024. |
| [53] | LI X, SONG X, KAMENECKA T M, et al.. Discovery of a highly selective CYP3A4 inhibitor suitable for reaction phenotyping studies and differentiation of CYP3A4 and CYP3A5[J]. Drug Metab. Dispos., 2012, 40(9): 1803-1809. |
| [54] | HU G, FANG Y, XU H, et al.. Identification of cytochrome P450 2E1 as a novel target in glioma and development of its inhibitor as an anti-tumor agent[J/OL]. Adv. Sci., 2023, 10(23): e2301096[2026-01-11]. . |
| [55] | ZHANG C, FANG Y, GUO M, et al.. Q11, a CYP2E1 inhibitor, exerts anti-hepatocellular carcinoma effect by inhibiting M2 macrophage polarization[J/OL]. Cancer Immunol. Immunother., 2024, 74(1): 35[2026-01-11]. . |
| [56] | ZHANG H, XU P, WANG T, et al.. Design, synthesis and biological evaluation of highly potent and selective CYP1B1 inhibitors[J]. New J. Chem., 2023, 47(12): 5680-5690. |
| [57] | HAKKOLA J, HUKKANEN J, TURPEINEN M, et al.. Inhibition and induction of CYP enzymes in humans: an update[J]. Arch. Toxicol., 2020, 94(11): 3671-3722. |
| [58] | HOPPE J M, HOLDERIED A, SCHÖNERMARCK U, et al.. Drug-induced CYP induction as therapy for tacrolimus intoxication[J]. Clin. Nephrol. Case Stud., 2022, 10: 42-46. |
| [59] | FUHR L M, MAROK F Z, HANKE N, et al.. Pharmacokinetics of the CYP 3A4 and CYP2B6 inducer carbamazepine and its drug-drug interaction potential: a physiologically based pharmacokinetic modeling approach[J/OL]. Pharmaceutics, 2021, 13(2): 270[2026-01-11]. . |
| [60] | LIU X Y, GUO Z T, CHEN Z D, et al.. Alflutinib (AST2818), primarily metabolized by CYP3A4, is a potent CYP3A4 inducer[J]. Acta Pharmacol. Sin., 2020, 41(10): 1366-1376. |
| [61] | YANG C G, CHEN T, SI W T, et al.. High-performance PBPK model for predicting CYP 3A4 induction-mediated drug interactions: a refined and validated approach[J/OL]. Front. Pharmacol., 2025, 16: 1521068[2026-01-11]. . |
| [62] | CHIANG M, SYCHTERZ C, PERERA V, et al.. Physiologically based pharmacokinetic modeling and simulation of mavacamten exposure with drug-drug interactions from CYP inducers and inhibitors by CYP2C19 phenotype[J]. Clin. Pharmacol. Ther., 2023, 114(4): 922-932. |
| [63] | ZHAI J, MAN V H, JI B, et al.. Comparison and summary of in silico prediction tools for CYP450-mediated drug metabolism[J/OL]. Drug Discov. Today, 2023, 28(10): 103728[2026-01-11]. . |
| [64] | WANG T, ZHAO L, LIU M, et al.. Oral intake of hydrogen-rich water ameliorated chlorpyrifos-induced neurotoxicity in rats[J]. Toxicol. Appl. Pharmacol., 2014, 280(1): 169-176. |
| [65] | 马雪梅,张鑫,谢飞,等.氢气生物学作用的生物酶基础[J].生物技术进展,2020,10(1):15-22. |
| MA X M, ZHANG X, XIE F, et al.. Bio-enzyme basis of hydrogen in biological system[J]. Curr. Biotechnol., 2020, 10(1): 15-22. | |
| [66] | 仪杨,张晓康,郭博远,等.氢气对辣根过氧化物酶活性的影响及其作用机制的研究[J].中国生物化学与分子生物学报,2020,36(7):811-819. |
| YI Y, ZHANG X K, GUO B Y, et al.. Effect of hydrogen on horseradish peroxidase activity and its mechanism[J]. Chin. J. Biochem. Mol. Biol., 2020, 36(7): 811-819. | |
| [67] | SUN Y, SUN P, GUO W. Fluorescent probes for iron, heme, and related enzymes[J/OL]. Coord. Chem. Rev., 2021, 429: 213645[2026-01-11]. . |
| [1] | 赵茹萍, 姜雪, 静悦, 段鑫源, 谢飞. 硫氧还蛋白系统的研究进展[J]. 生物技术进展, 2026, 16(1): 18-28. |
| [2] | 王惠, 赵鹏翔, 张旭娟, 姚婷婷, 刘梦昱, YAO Mawulikplimi Adzavon, 谢飞. 间充质干细胞在疾病治疗中的应用潜力[J]. 生物技术进展, 2021, 11(6): 688-693. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||