


生物技术进展 ›› 2026, Vol. 16 ›› Issue (1): 18-28.DOI: 10.19586/j.2095-2341.2025.0127
收稿日期:2025-09-23
接受日期:2025-11-04
出版日期:2026-01-25
发布日期:2026-02-12
通讯作者:
谢飞
作者简介:赵茹萍 E-mail: Zhaoruping@emails.bjut.edu.cn;
基金资助:
Ruping ZHAO( ), Xue JIANG, Yue JING, Xinyuan DUAN, Fei XIE(
), Xue JIANG, Yue JING, Xinyuan DUAN, Fei XIE( )
)
Received:2025-09-23
Accepted:2025-11-04
Online:2026-01-25
Published:2026-02-12
Contact:
Fei XIE
摘要:
硫氧还蛋白系统是机体内重要的抗氧化和还原系统,主要由硫氧还蛋白、硫氧还蛋白还原酶以及辅因子烟酰胺腺嘌呤二核苷酸磷酸构成。该系统在维持细胞内氧化还原平衡、调控信号转导、调节细胞增殖和凋亡等方面发挥关键作用。近年来,大量研究揭示了硫氧还蛋白系统在多种疾病发生发展中的作用,为相关疾病的治疗提供了新的靶点和策略。综述了硫氧还蛋白系统的分子结构、作用机制、在生理及病理状态下的功能,以及其作为药物靶点的潜在应用,并对未来研究方向进行了展望,为深入理解其在生理病理过程中的作用机制,以及相关疾病的靶向治疗提供理论依据和新的思路。
中图分类号:
赵茹萍, 姜雪, 静悦, 段鑫源, 谢飞. 硫氧还蛋白系统的研究进展[J]. 生物技术进展, 2026, 16(1): 18-28.
Ruping ZHAO, Xue JIANG, Yue JING, Xinyuan DUAN, Fei XIE. The Thioredoxin System: Recent Progress and Developments[J]. Current Biotechnology, 2026, 16(1): 18-28.
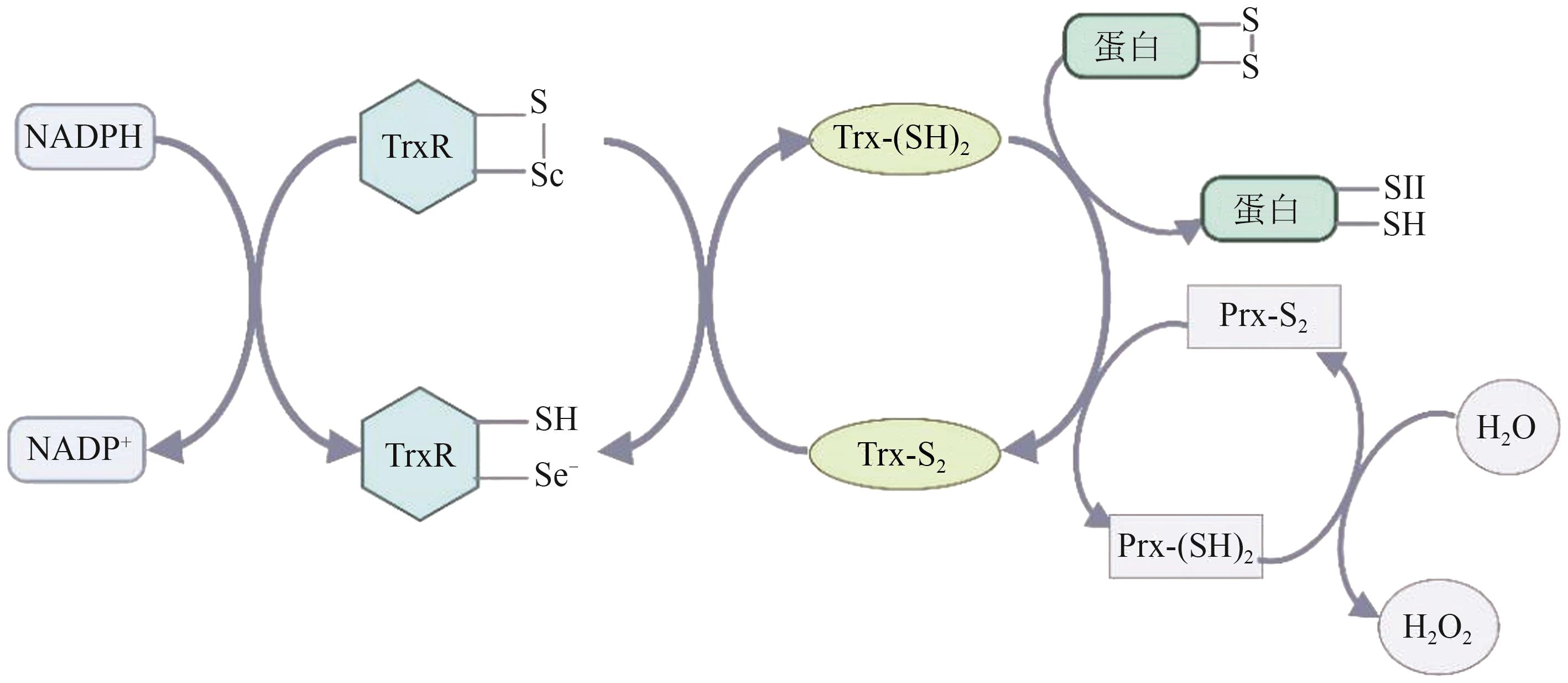
图1 硫氧还蛋白系统的组成[1]注:NADPH—还原型烟酰胺腺嘌呤二核苷酸磷酸;NADP+—氧化型烟酰胺腺嘌呤二核苷酸磷酸;TrxR—硫氧还蛋白还原酶;Trx-(SH)2—还原型硫氧还蛋白;Trx-S2—氧化型硫氧还蛋白;Prx-S2—氧化型过氧化物酶;Prx-(SH)2—还原型过氧化物酶;H2O2—过氧化氢。
Fig. 1 Components of the thioredoxin systeme[1]
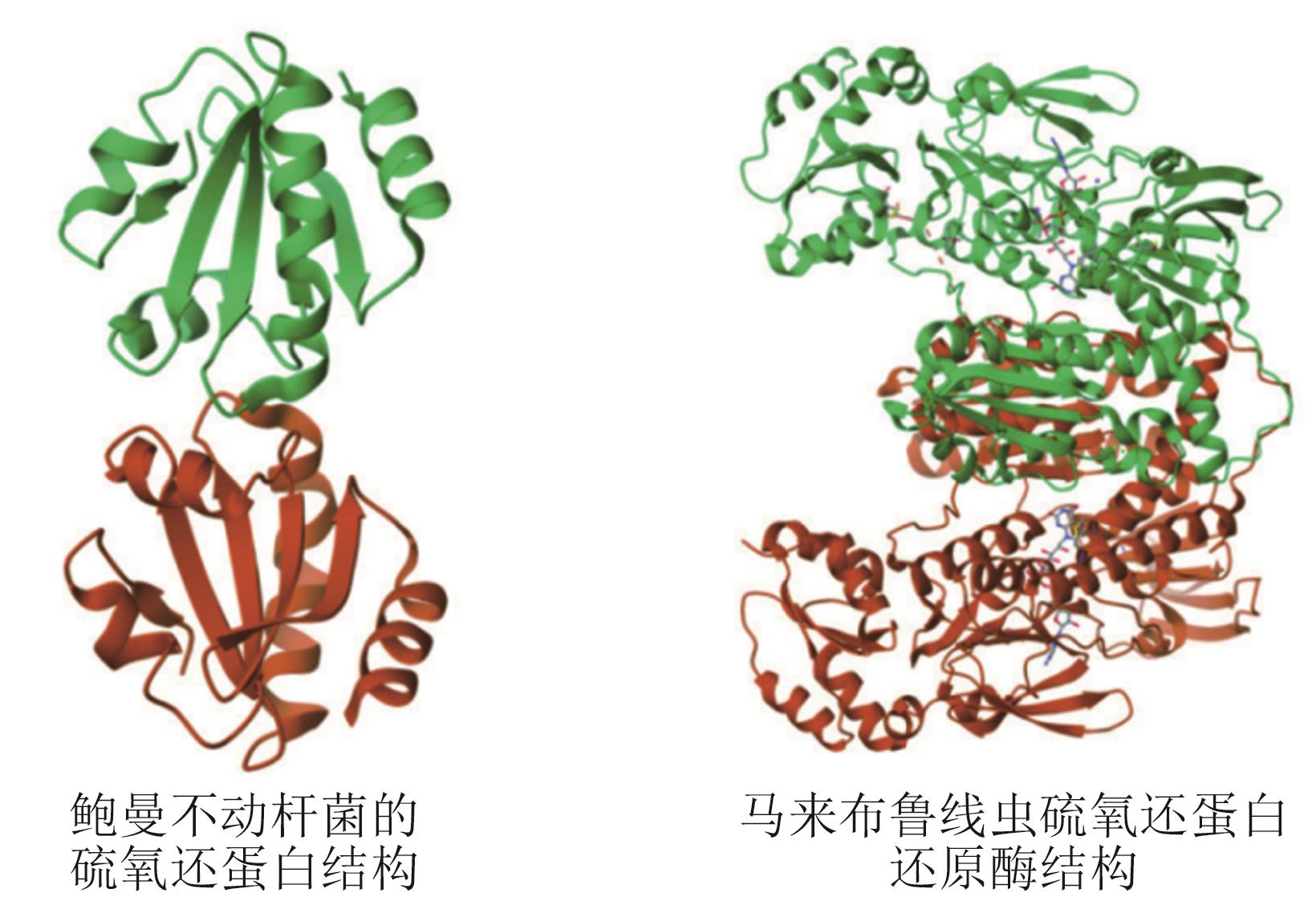
图2 鲍曼不动杆菌硫氧还蛋白[3]及马来布鲁线虫硫氧还蛋白还原酶[4]的结构示意图
Fig. 2 Structures representations of Acinetobacter baumannii thioredoxin and Brugia malayi thioredoxin reductase
表1 硫氧还蛋白系统抑制剂的分类
Table 1 Classification of thioredoxin system inhibitors
| [1] | BJØRKLUND G, ZOU L, WANG J, et al.. Thioredoxin reductase as a pharmacological target[J/OL]. Pharmacol. Res., 2021, 174: 105854[2025-09-02]. . |
| [2] | FRITZ-WOLF K, KEHR S, STUMPF M, et al.. Crystal structure of the human thioredoxin reductase-thioredoxin complex[J/OL]. Nat. Commun., 2011, 2: 383[2025-09-02]. . |
| [3] | CHANG Y J, PARK H H. High-resolution crystal structure of Acinetobacter baumannii thioredoxin 1[J]. Biochem. Biophys. Res. Commun., 2022, 608: 1-7. |
| [4] | FATA F, GENCHEVA R, CHENG Q, et al.. Biochemical and structural characterizations of thioredoxin reductase selenoproteins of the parasitic filarial nematodes Brugia malayi and Onchocerca volvulus [J/OL]. Redox Biol., 2022, 51: 102278[2025-09-02]. . |
| [5] | BJÖRNBERG O, VIENNET T, SKJOLDAGER N, et al.. Lactococcus lactis thioredoxin reductase is sensitive to light inactivation[J]. Biochemistry, 2015, 54(8): 1628-1637. |
| [6] | 王全富,苗苗,侯艳华,等.硫氧还蛋白研究进展[J].生物技术进展,2015,5(3):196-200. |
| WANG Q F, MIAO M, HOU Y H, et al.. Progress on thioredoxin[J]. Curr. Biotechnol., 2015, 5(3): 196-200. | |
| [7] | STOMBERSKI C T, HESS D T, STAMLER J S. Protein S-nitrosylation: determinants of specificity and enzymatic regulation of S-nitrosothiol-based signaling[J]. Antioxid. Redox Signal., 2019, 30(10): 1331-1351. |
| [8] | TANAKA T. Thioredoxin-2 (TRX-2) is an essential gene regulating mitochondria-dependent apoptosis[J]. EMBO J., 2002, 21(7): 1695-1703. |
| [9] | MIRANDA-VIZUETE A, LJUNG J, DAMDIMOPOULOS A E, et al.. Characterization of sptrx, a novel member of the thioredoxin family specifically expressed in human spermatozoa[J]. J. Biol. Chem., 2001, 276(34): 31567-31574. |
| [10] | MAIA L B, MAITI B K, MOURA I, et al.. Selenium: more than just a fortuitous sulfur substitute in redox biology[J/OL]. Molecules, 2024, 29(1): 120[2025-09-02]. . |
| [11] | ARNÉR E S J. Focus on mammalian thioredoxin reductases: important selenoproteins with versatile functions[J]. Biochim. Biophys. Acta, 2009, 1790(6): 495-526. |
| [12] | VESZELYI K, CZEGLE I, VARGA V, et al.. Subcellular localization of thioredoxin/thioredoxin reductase system: a missing link in endoplasmic reticulum redox balance[J/OL]. Int. J. Mol. Sci., 2024, 25(12): 6647[2025-09-02]. . |
| [13] | FAN J, YE J, KAMPHORST J J, et al.. Quantitative flux analysis reveals folate-dependent NADPH production[J]. Nature, 2014, 510(7504): 298-302. |
| [14] | KIM D, KESAVAN R, RYU K, et al.. Mitochondrial NADPH fuels mitochondrial fatty acid synthesis and lipoylation to power oxidative metabolism[J]. Nat. Cell Biol., 2025, 27(5): 790-800. |
| [15] | HASAN AALI, KALININA E, TATARSKIY V, et al.. The thioredoxin system of mammalian cells and its modulators[J/OL]. Biomedicines, 2022, 10(7): 1757[2025-09-02]. . |
| [16] | LU M, JI J, LV Y, et al.. Bivalent inhibitors of the BTB E3 ligase KEAP1 enable instant NRF2 activation to suppress acute inflammatory response[J]. Cell Chem. Biol., 2024, 31(6): 1188-1202. |
| [17] | HE X, MA Q. Redox regulation by nuclear factor erythroid 2-related factor 2: gatekeeping for the basal and diabetes-induced expression of thioredoxin-interacting protein[J]. Mol. Pharmacol., 2012, 82(5): 887-897. |
| [18] | HAN Y Y, GU X, YANG C Y, et al.. Protective effect of dimethyl itaconate against fibroblast-myofibroblast differentiation during pulmonary fibrosis by inhibiting TXNIP[J]. J. Cell. Physiol., 2021, 236(11): 7734-7744. |
| [19] | GUEVARA-FLORES A, MARTÍNEZ-GONZÁLEZ J D J, RENDÓN J L, et al.. The architecture of thiol antioxidant systems among invertebrate parasites[J]. Antioxidants, 2017, 6(2): 39[2025-09-02]. . |
| [20] | KELLEHER Z T, SHA Y, FOSTER M W, et al.. Thioredoxin-mediated denitrosylation regulates cytokine-induced nuclear factor κB (NF-κB) activation[J]. J. Biol. Chem., 2014, 289(5): 3066-3072. |
| [21] | CHEN B, GUAN D, CUI Z J, et al.. Thioredoxin 1 downregulates MCP-1 secretion and expression in human endothelial cells by suppressing nuclear translocation of activator protein 1 and redox factor-1[J]. Am. J. Physiol. Cell Physiol., 2010, 298(5): 1170-1179. |
| [22] | SHAO D, OKA S I, LIU T, et al.. A redox-dependent mechanism for regulation of AMPK activation by Thioredoxin1 during energy starvation[J]. Cell Metab., 2014, 19(2): 232-245. |
| [23] | 姜晓宇,陈异,倪晶晶.miR-93通过激活PI3K/AKT通路调节免疫介导创伤性脑损伤的机制研究[J].生物技术进展,2025,15(4):711-719. |
| JIANG X Y, CHEN Y, NI J J. Mechanism of miR-93 regulating immune-mediated traumatic brain injury through activation of PI3K/AKT pathway[J]. Curr. Biotechnol., 2025, 15(4): 711-719. | |
| [24] | MEUILLET E J, MAHADEVAN D, BERGGREN M, et al.. Thioredoxin-1 binds to the C2 domain of PTEN inhibiting PTEN's lipid phosphatase activity and membrane binding: a mechanism for the functional loss of PTEN's tumor suppressor activity[J]. Arch. Biochem. Biophys., 2004, 429(2): 123-133. |
| [25] | YANG L, ZENG C, ZHANG Y, et al.. Functions of thioredoxin1 in brain development and in response to environmental chemicals in zebrafish embryos[J]. Toxicol. Lett., 2019, 314: 43-52. |
| [26] | SIMÃO S, AGOSTINHO R R, MARTÍNEZ-RUIZ A, et al.. Regulation of ras signaling by S-nitrosylation[J/OL]. Antioxidants, 2023, 12(8): 1562[2025-09-02]. . |
| [27] | CHATTERJI A, SACHIN K, SENGUPTA R. Glutathione-dependent thioredoxin reduction and lipoamide system support in-vitro mammalian ribonucleotide reductase catalysis: a possible antioxidant redundancy[J]. Mol. Biol. Rep., 2022, 49(8): 8179-8183. |
| [28] | KOC A, MATHEWS C K, WHEELER L J, et al.. Thioredoxin is required for deoxyribonucleotide pool maintenance during S phase[J]. J. Biol. Chem., 2006, 281(22): 15058-15063. |
| [29] | LEE S J, TRAN N Q, LEE J, et al.. Hydrophobic residue in Escherichia coli thioredoxin critical for the processivity of T7 DNA polymerase[J]. Biochemistry, 2018, 57(40): 5807-5817. |
| [30] | GHOSH S, HAMDAN S M, COOK T E, et al.. Interactions of Escherichia coli thioredoxin, the processivity factor, with bacteriophage T7 DNA polymerase and helicase[J]. J. Biol. Chem., 2008, 283(46): 32077-32084. |
| [31] | MURI J, HEER S, MATSUSHITA M, et al.. The thioredoxin-1 system is essential for fueling DNA synthesis during T-cell metabolic reprogramming and proliferation[J]. Nat. Commun., 2018, 9: 5391[2025-09-02]. . |
| [32] | MCMANUS R M, LATZ E. NLRP3 inflammasome signalling in Alzheimer's disease[J/OL]. Neuropharmacology, 2024, 252: 109941[2025-09-02]. . |
| [33] | FARKHONDEH T, FOLGADO S L, POURBAGHER-SHAHRI A M, et al.. The therapeutic effect of resveratrol: focusing on the Nrf2 signaling pathway[J/OL]. Biomed. Pharmacother., 2020, 127: 110234[2025-09-02]. . |
| [34] | 谢婧雯,赵艳,奉夏露,等.丁苯酞对Aβ25-35诱导的阿尔茨海默病细胞模型的保护作用[J].南方医科大学学报,2017,37(9):1228-1233. |
| XIE J W, ZHAO Y, FENG X L, et al.. Protective effect of butylphthalide in a cell model of Alzheimer's disease induced by Aβ25-35 in Neuro 2a cells[J]. J. South. Med. Univ., 2017, 37(9): 1228-1233. | |
| [35] | LIU Z, YE Q, WANG F, et al.. Protective effect of thioredoxin reductase 1 in Parkinson's disease[J]. Neurosci. Lett., 2021,741: 135457[2025-09-02]. . |
| [36] | ZHANG X, DENG R, ZHANG S, et al.. Thioredoxin-1 regulates calcium homeostasis in MPP(+)/MPTP-induced Parkinson's disease models[J]. Eur. J. Neurosci., 2021, 54(3): 4827-4837. |
| [37] | LIN K J, CHEN S D, LIN K L, et al.. Iron brain menace: the involvement of ferroptosis in Parkinson disease[J/OL]. Cells, 2022, 11(23): 3829[2025-09-02]. . |
| [38] | LIU Z, YE Q, WANG F, et al.. Overexpression of thioredoxin reductase 1 can reduce DNA damage, mitochondrial autophagy and endoplasmic reticulum stress in Parkinson’s disease[J]. Exp. Brain Res., 2021, 239(2): 475-490. |
| [39] | AL-HADYAN K S, STORR S J, ZAITOUN A M, et al.. Thioredoxin system protein expression in carcinomas of the pancreas, distal bile duct, and Ampulla in the United Kingdom[J/OL]. Diseases, 2024, 12(10): 227[2025-09-02]. . |
| [40] | DENG J, PAN T, LIU Z, et al.. The role of TXNIP in cancer: a fine balance between redox, metabolic, and immunological tumor control[J]. Br. J. Cancer, 2023, 129(12): 1877-1892. |
| [41] | ZOU Q, CHEN Y F, ZHENG X Q, et al.. Novel thioredoxin reductase inhibitor butaselen inhibits tumorigenesis by down-regulating programmed death-ligand 1 expression[J]. J. Zhejiang Univ. Sci. B, 2018, 19(9): 689-698. |
| [42] | 李仪扬,周执政,王淑菲,等.CRISPR/Cas9基因编辑技术在疾病治疗中的应用与展望[J].生物技术进展,2025,15(1):35-42. |
| LI Y Y, ZHOU Z Z, WANG S F, et al.. Application and prospect of CRISPR/Cas9 gene editing technology in disease treatment[J]. Curr. Biotechnol., 2025, 15(1): 35-42. | |
| [43] | TINKOV A A, BJØRKLUND G, SKALNY A V, et al.. The role of the thioredoxin/thioredoxin reductase system in the metabolic syndrome: towards a possible prognostic marker?[J]. Cell. Mol. Life Sci., 2018, 75(9): 1567-1586. |
| [44] | DOMINGUES A, BOISSON-VIDAL C, MARQUET DE ROUGE P, et al.. Targeting endothelial thioredoxin-interacting protein (TXNIP) protects from metabolic disorder-related impairment of vascular function and post-ischemic revascularisation[J]. Angiogenesis, 2020, 23(2): 249-264. |
| [45] | FONSECA-PEREIRA P D A, SOUZA P V L, FERNIE A R, et al.. Thioredoxin-mediated regulation of (photo)respiration and central metabolism[J]. J. Exp. Bot., 2021, 72(17): 5987-6002. |
| [46] | CHAKRABORTY P, CHATTERJEE S, KESARWANI P, et al.. Thioredoxin-1 improves the immunometabolic phenotype of antitumor T cells[J]. J. Biol. Chem., 2019, 294(23): 9198-9212. |
| [47] | KARUNANITHI S, LIU R, HOU Y, et al.. Thioredoxin reductase is a major regulator of metabolism in leukemia cells[J]. Oncogene, 2021, 40(33): 5236-5246. |
| [48] | LÓPEZ-GRUESO M J, GONZÁLEZ-OJEDA R, REQUEJO-AGUILAR R, et al.. Thioredoxin and glutaredoxin regulate metabolism through different multiplex thiol switches[J/OL]. Redox Biol., 2019, 21: 101049[2025-09-02]. . |
| [49] | PARK S H, LEE J H, BEREK J S, et al.. Auranofin displays anticancer activity against ovarian cancer cells through FOXO3 activation independent of p53[J]. Int. J. Oncol., 2014, 45(4): 1691-1698. |
| [50] | PICKERING I J, CHENG Q, RENGIFO E M, et al.. Direct observation of methylmercury and auranofin binding to selenocysteine in thioredoxin reductase[J]. Inorg. Chem., 2020, 59(5): 2711-2718. |
| [51] | CHEN J C, ZHANG Y, JIE X M, et al.. Ruthenium(Ⅱ) salicylate complexes inducing ROS-mediated apoptosis by targeting thioredoxin reductase[J]. J. Inorg. Biochem., 2019, 193: 112-123. |
| [52] | ZHANG J J, MUENZNER J K, MAATY M, et al.. A multi-target caffeine derived rhodium(i) N-heterocyclic carbene complex: evaluation of the mechanism of action[J]. Dalton Trans., 2016, 45(33): 13161-13168. |
| [53] | WANG L, YANG Z, FU J, et al.. Ethaselen: a potent mammalian thioredoxin reductase 1 inhibitor and novel organoselenium anticancer agent[J]. Free Radic. Biol. Med., 2012, 52(5): 898-908. |
| [54] | SHE W, SHI X, LIU T, et al.. Discovery of novel organoarsenicals as robust thioredoxin reductase inhibitors for oxidative stress mediated cancer therapy[J/OL]. Biochem. Pharmacol., 2023, 218: 115908[2025-09-02]. . |
| [55] | ZHANG D, LIU Y, LUO Z, et al.. The novel thioredoxin reductase inhibitor A-Z2 triggers intrinsic apoptosis and shows efficacy in the treatment of acute myeloid leukemia[J]. Free Radic. Biol. Med., 2020, 146: 275-286. |
| [56] | FEKY S E, ABDEL G M M, MONEIM N, et al.. Cytotoxic, chemosensitizing and radiosensitizing effects of curcumin based on thioredoxin system inhibition in breast cancer cells: 2D vs. 3D cell culture system[J/OL]. Exp. Ther. Med., 2021, 21(5): 506[2025-09-02]. . |
| [57] | ZHANG Y, SUN S, XU W, et al.. Thioredoxin reductase 1 inhibitor shikonin promotes cell necroptosis via SecTRAPs generation and oxygen-coupled redox cycling[J]. Free Radic. Biol. Med., 2022, 180: 52-62. |
| [58] | YANG Y, SUN S, XU W, et al.. Piperlongumine inhibits thioredoxin reductase 1 by targeting selenocysteine residues and sensitizes cancer cells to erastin[J]. Antioxidants, 2022,11(4): 710[2025-09-02]. . |
| [59] | DUAN D, ZHANG B, YAO J, et al.. Gambogic acid induces apoptosis in hepatocellular carcinoma SMMC-7721 cells by targeting cytosolic thioredoxin reductase[J]. Free. Radic. Biol. Med., 2014, 69: 15-25. |
| [60] | JOVANOVIĆ M, ZHUKOVSKY D, PODOLSKI-RENIĆ A, et al.. Further exploration of DVD-445 as a lead thioredoxin reductase (TrxR) inhibitor for cancer therapy: optimization of potency and evaluation of anticancer potential[J/OL]. Eur. J. Med. Chem., 2020, 191: 112119[2025-09-02]. . |
| [61] | TULADHAR A, REIN K S. Manumycin a is a potent inhibitor of mammalian thioredoxin reductase-1 (TrxR-1)[J]. ACS Med. Chem. Lett., 2018, 9(4): 318-322. |
| [62] | WANG C, LI S, ZHAO J, et al.. Design and SAR of withangulatin a analogues that act as covalent TrxR inhibitors through the Michael addition reaction showing potential in cancer treatment[J]. J. Med. Chem., 2020, 63(19): 11195-11214. |
| [63] | NEMEIKAITĖ-ČĖNIENĖ A, MISEVIČIENĖ L, MAROZIENĖ A, et al.. Enzymatic redox properties and cytotoxicity of irreversible nitroaromatic thioredoxin reductase inhibitors in mammalian cells[J/OL]. Int. J. Mol. Sci., 2023, 24(15): 12460[2025-09-02]. . |
| [64] | CENAS N, PRAST S, NIVINSKAS H, et al.. Interactions of nitroaromatic compounds with the mammalian selenoprotein thioredoxin reductase and the relation to induction of apoptosis in human cancer cells[J]. J. Biol. Chem., 2006, 281(9): 5593-5603. |
| [65] | PETUKHOVA V Z, ABOAGYE S Y, ARDINI M, et al.. Non-covalent inhibitors of thioredoxin glutathione reductase with schistosomicidal activity in vivo [J/OL]. Nat. Commun., 2023, 14(1): 3737[2025-09-02]. . |
| [66] | ZHOU X, NIE Q, LIU Q, et al.. Discovery of flavopiridol as a noncovalent thioredoxin reductase inhibitor through in silico and in vitro approach[J/OL]. Int. J. Biol. Macromol., 2025, 311(Pt 2): 143641[2025-09-02]. . |
| [67] | LUNDBERG M, MATTSSON Å, REISER K, et al.. Inhibition of the thioredoxin system by PX-12 (1-methylpropyl 2-imidazolyl disulfide) impedes HIV-1 infection in TZM-bl cells[J/OL]. Sci. Rep., 2019, 9(1): 5656[2025-09-02]. . |
| [68] | EHRENFELD V, FULDA S. Thioredoxin inhibitor PX-12 induces mitochondria-mediated apoptosis in acute lymphoblastic leukemia cells[J]. Biol. Chem., 2020, 401(2): 273-283. |
| [69] | BAKER A F, ADAB K N, RAGHUNAND N, et al.. A phase IB trial of 24-hour intravenous PX-12, a thioredoxin-1 inhibitor, in patients with advanced gastrointestinal cancers[J]. Invest. New Drugs, 2013, 31(3): 631-641. |
| [1] | 王惠, 赵鹏翔, 张旭娟, 姚婷婷, 刘梦昱, YAO Mawulikplimi Adzavon, 谢飞. 间充质干细胞在疾病治疗中的应用潜力[J]. 生物技术进展, 2021, 11(6): 688-693. |
| [2] | 张文颖,,王思情,张新妍,李庆伟,,李莹莹,. JAK2/STAT3作为新型抗癌药物靶点的研究进展[J]. 生物技术进展, 2021, 11(1): 33-39. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||